Pig heart transplanted to human for the first time
Companies, agencies, institutions, etc
the University of Maryland School of Medicine
the Food and Drug Administration
Revivicor
United Therapeutics
ClinicalTrials.gov
The University of Maryland's
the Ars Orbital Transmission
CNMN Collection
WIRED Media Group
Condé Nast
People
John Timmer
Ars
Groups
No matching tags
Physical locations
No matching tags
Places
No matching tags
Locations
No matching tags
Events
No matching tags
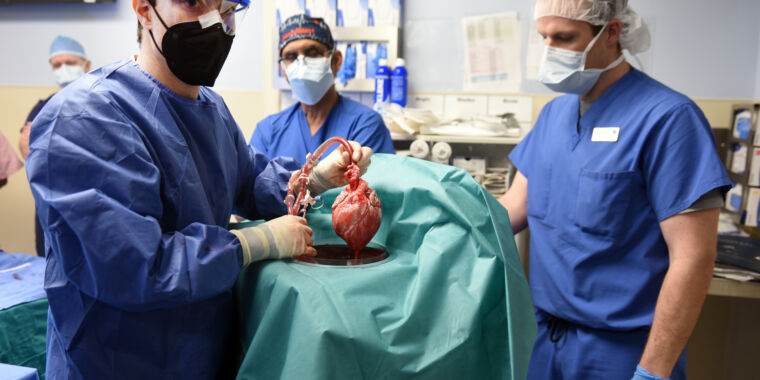
Summary
But the recent surgery wasn't part of a clinical trial, so it shouldn't be viewed as an indication that this approach is ready for widespread safety and efficacy testing.Instead, the surgery was authorized by the Food and Drug Administration under its "compassionate use" access program, which allows patients facing life-threatening illnesses to receive investigational treatments that haven't gone through rigorous clinical testing yet.The heart used for this transplant did come from a genetically modified line that was specifically engineered to reduce the chance of rejection by the human immune system. While it's easy to speculate on what these genes might be, the potential list of targets is much larger than what has actually been edited.Given the amount of effort that has gone into generating the pig lines, it was clearly a matter of time before these sorts of transplants were attempted.
As said here by John Timmer